Draw The Lewis Structure Of C2h4
Be sure to include all resonance structures that satisfy the octet rule. Is it polar or nonpolar.
Ethene C2h4 Lewis Structure Hybridization
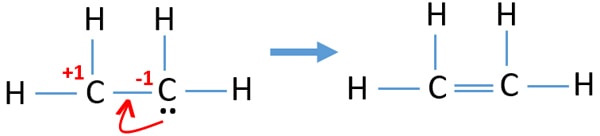
In a double bond two pairs of valence electrons are shared for a total of four valence electrons.

Draw the lewis structure of c2h4. Be sure to include all resonance structures that satisfy the octet rule. Can I draw resonance structures of C 2 H 4 from the lewis structure of C 2 H 4. It shows the bonding between the atoms of a molecule and it also shows the unpaired electrons present in the molecule.
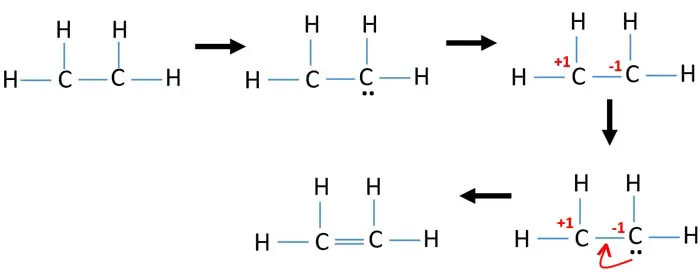
Count total valence electron in C2H4. Arrangement of atoms shown below dashed lines show connections between atoms. Carbon has 4 and hydrogen has 1 making a grand total of 12 e- 42 41.
It consists of two carbon molecules and 4 hydrogen molecules. Initial structure will appear as. Calculate the total valence electrons in the molecule.
The lewis structure of C2H4 is very easy to draw-Some steps need to follow for drawing the C2H4 Lewis dot structure 1. What is its molecular shape. To draw the c2h4 lewis.
Lewis dot structure of C 2 H 4. Alternatively a dot method can be used to draw the lewis structure. C2h4 Lewis Dot Structure - How to draw the lewis structure for c2h4.
By signing up youll get thousands of. But seriously you have an electron pair between the C and each of the Hs in the Lewis diagram a ala. Draw the Lewis structure of C2H4 and identify its geometry.
Draw the Lewis structure for the ethylene C2H4 molecule. No lone pair is present on the central or outer atom in the lewis structure of ethene. Draw the Lewis structure for ethylene C2H4.
Drawing the Lewis structure for C. Valence electrons of C is 4 while H is 1. Lewis Dot Structure for C2H4 6 of 6 Watch the video of Dr.
Draw the Lewis structure for the ethylene C2H4 molecule. Electron Dot Structure for ethene C2H4. 6 rows For C 2 H 4 you have a total of 12 total valence electrons.
To know whether resonance structures can be drawn for C 2 H 4 you should understand the structure of lewis structure of C 2 H 4. Lewis benzene dot 3. Dot is used to show the valance electrons.
Such as polarity and bond length we will draw the lewis or electron dot structure. Use information from step 4 and 5 to draw the lewis structure. Draw the electron-dot structure for C2H4.
I would be lazy and look it up on the internet. There are only single bond between carbon atom and hydrogen atom because. The electron dot structure is drawn using Lewis-dot structure.
Is 12 4 2 1 4 12 Following the skeletal formula structure will appear as C being attached to each other with 2 Hs on it. To draw the Lewis structure for C2H4 the total number of valence electrons must be known. Drawing the Lewis structure for C 2 H 4 named ethene requires the use of a double bond.
C2H4 Lewis structure contains four C-H bonds and one double bond in between two carbon atoms. For C 2 H 4 you have a total of 12 total valence electrons. Drawing the Lewis Structure for C 2 H 2.
By signing up youll get thousands of step-by-step solutions to your. Draw the Lewis structure for C2H4. H-----C---H SHOW WORK This question is worth a total of 6 points.
Welcome to Geometry of Molecules and today in this video we are going to help you know the step-by-step method for determining the Lewis Structure. A step-by-step explanation of how to draw the C2H2 Lewis Dot Structure Ethyne or AcetyleneFor the C2H2 structure use the periodic table to find the total. The key to understanding how t.
All electrons are shared within bonds in this compound. A step-by-step explanation of how to write the Lewis Dot Structure for C2H4 ethene. Drawing the Lewis dot structure for C2H4 ethene and answer the questions below.
Ortho-dichlorobenzene C6H4Cl2 is obtained when two of the adjacent hydrogen atoms in benzene are replaced with Cl atoms. 1 point for the correct selections assessed when you answer and 5 points for the Lewis structure on your work assessed when I review. Note that the C 2 H 4 Lewis dot structure involves sharing more than one pair of electrons.
In the lewis structure of C 2 H 4 there are only four C-H bonds one CC bond and no lone pairs on last shells. When there are no polar bonds in a molecule there is no permanent charge difference between one part of the molecule and another and a.
Ethene C2h4 Lewis Structure Hybridization

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist

Draw The Lewis Structure For The C2h4 Ske Clutch Prep
Lewis Structure Of C2h6 Ethane Youtube
Lewis Electron Dot Structures Ck 12 Foundation

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
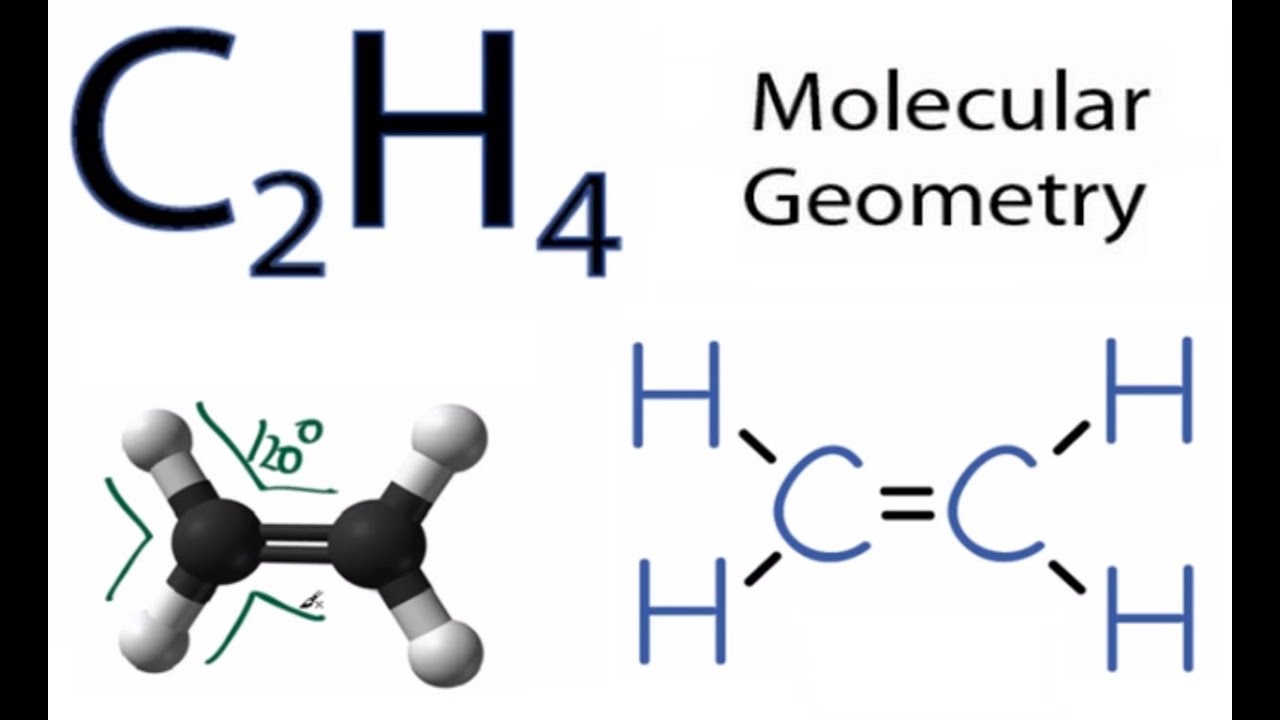
C2h4 Molecular Geometry Shape And Bond Angles Youtube
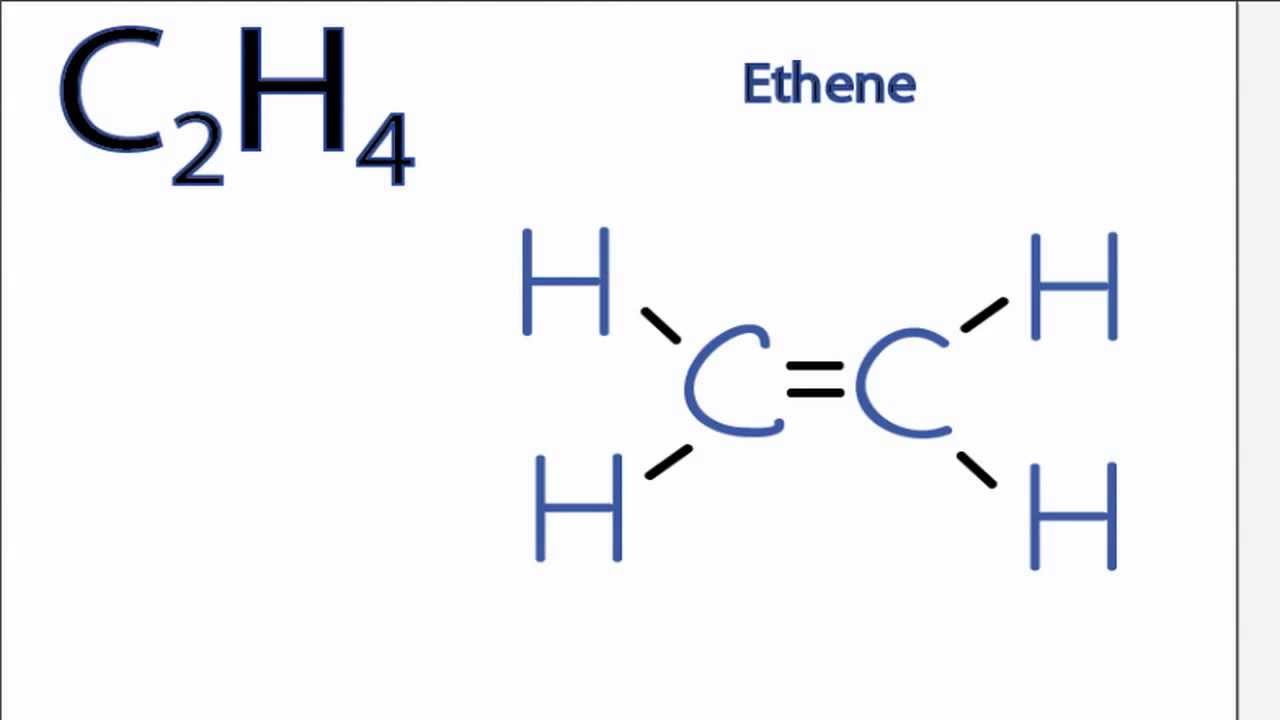
Is C2h4 Polar Or Nonpolar Youtube

Draw The Electron Dot Structure Of Ethene C2h4 Brainly In

C2h4 Lewis Structure C2h4 Lewis Structure Molecular Geometry

C2h4 Lewis Structure Molecular Geometry Hybridization And Mo Diagram Techiescientist
Lewis Structure Of C2h4 Biochemhelp
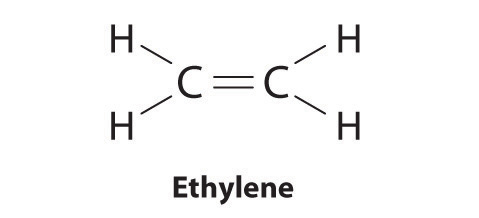
10 4 Writing Lewis Structures Chemistry Libretexts

Multiple Covalent Bonds Chemistry For Non Majors
Ethene C2h4 Lewis Structure Hybridization
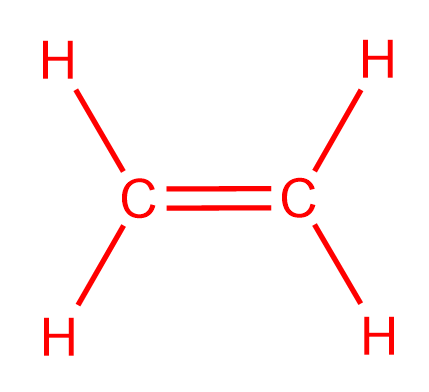
Draw The Lewis Structure For The C2h4 Ske Clutch Prep

Draw The Lewis Structure For Ethylene C2h Clutch Prep

Write Lewis Structure Of C2h4 Brainly In

