Electron Dot Formula For Ni3
States vary between high-spin and low-spin configurations sure you can figure out how ce. Try using one double bond.

25 8 2013 18 8 22 1 Ensenanza De Quimica Quimica Notas De Quimica
CHEMICAL AND PHYSICAL INFORMATION - ATSDR Home 3.
Electron dot formula for ni3. It can even be detonated by alpha radiation. NI3 Lewis Structure - How to Draw the Dot Structure for NI3 Nitrogen Triiodide - YouTube. MATERIAL SAFETY DATA SHEET - Seton Resource Center Formula.
The octet rule is satisfied but we have too many electrons. Answer to Draw the electron-dot formula for each of the following moleculesa. E Dot Formula Of Ni3.
How to Draw the Lewis Structure for Draw the electron dot structure of Nitrogen molecule N2O Molecular Geometry Shape and Bond Angles YouTube. Nickel ion Ni3 UNII-J7B7WZ5ZMT. Small quantities explode with a loud sharp snap when touched even lightly releasing a purple cloud of iodine vapor.
Nitrogen is a chemical element with symbol n and atomic number 7. The lewis dot structure of a. N2 Electron Dot Formula Hydrogen Bonding Chemistry for Non Majors N2H2 Lewis Structure.
Tp15-c4pdf - Read File Online - Report Abuse. In N 2 one nitrogen atom shares its three electrons with another nitrogen atom to form a covalent. Lets do the Lewis Structure for NI3 Nitrogen Triiodide.
N has five valence electrons 3 of which are going to be shared with the iodines since there are 3 iodines which leaves you with two left over. For structure calculate total number valence electrons the. It is the least.
Log in Sign up. Geometry gives the spatial arrangement of electron pairs can be either bonding electrons or non-bonding electrons around central. October 4th 2013 021818 AM.
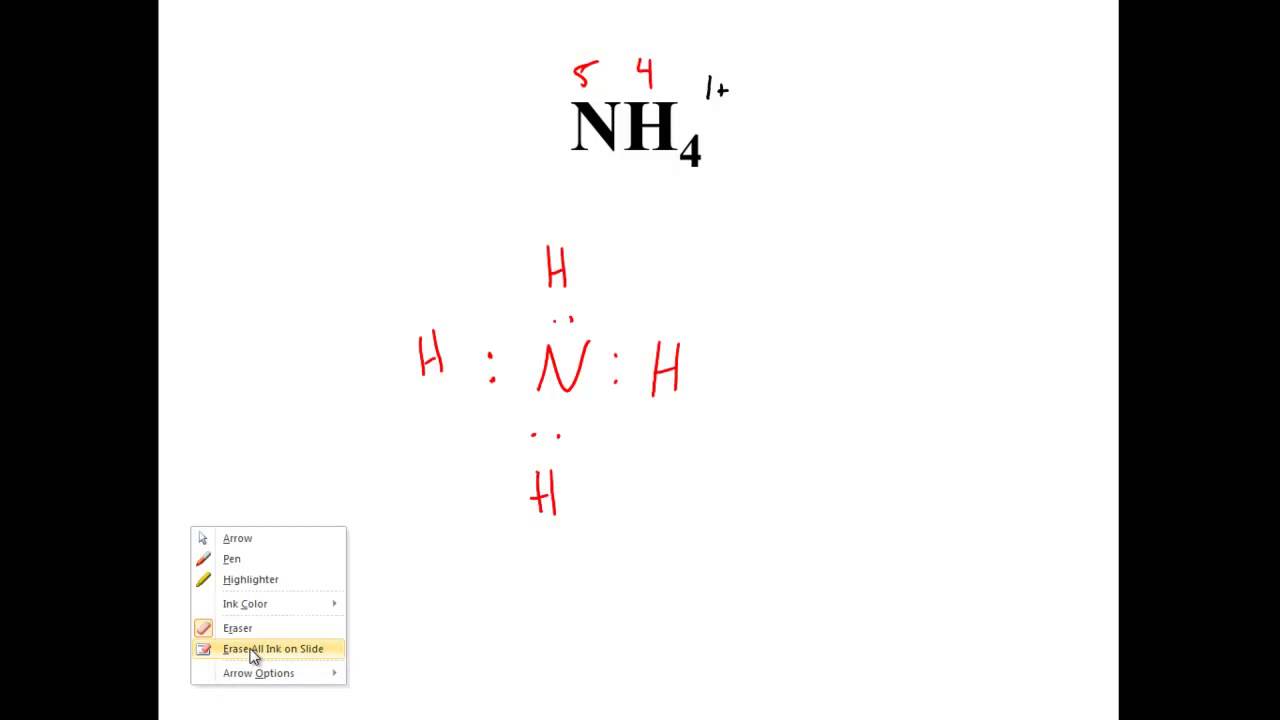
A Lewis structure also called Lewis dot formulas Lewis dot structures or electron dot structures are pictorial diagrams that represent the bonding between atoms in a compound and the placement of electrons. And then for Iodine we have seven valence electrons but theres three of them so 5 21 26 total valence electrons. That makes three lone electrons from nitrogen as a total one of each three being shared with one of the other electrons.
Bonds and thus has 4 valence electrons s ni3 electron group arrangement electrons thus 4. Formula Chemical Ni u2013 O. CAS Chemical Abstracts Service.
Answer and Explanation. Are molecular or coval. Nitrogen iodide NI3 6CI7CI8CI9CI More.
Nitrogen is in Group 5 sometimes called fifteen so it has five valence electrons. Electron-dot structure of N 2 is. So lets look on the periodic table.
Lewis dot structures for polyatomic ions this video shows how to draw ions. 18 electrons fill up the third electron shell leaving 10 valance electrons 3 4 it five. Nitrogen triiodide is an inorganic compound with the formula N I 3.
Now fill in enough pairs of electrons to get an octet of electrons around each atom. Therefore the total number of valence electrons is 26. OSO And then count the total number of valence electrons in the structure.
There are 7 valence electrons in an iodine atom and five electrons in nitrogen. Ni3 Lewis Structure How To Draw The Dot. The structure must have a total of 8 valence electrons because there are 2.
It is an extremely sensitive contact explosive. What is the electron dot formula for. A stepbystep explanation of how to draw the ni3 lewis dot structure nitrogen triiodide.
Therefore there are 20 valence electrons remaining. DOTUNNAIMCO Department of TransportationUnited NationsNorth Filename. A step by step explanation of how to draw the no lewis structure nitric oxide.
Electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the elements symbol. Big explosion nice purple cloud. 2 bonding pairs 8 nonbonding pairs 10 electron pairs total which is 20 valence electrons in the structure two too many.
Step 3 A pair of bonding electrons is placed between each I atom and central N atom. So youd have the N with three iodines attached to it sharing one of their seven valence electrons with one of the electrons from nitrogen. That is exactly what is done to get the correct lewis structure for nitrogen monoxide.
At the same time Lewis electron dot formula is taken more powerful and descriptive in nature and makes the calculation little easier as compared to other techniques available so far. Lets put Nitrogen in the center. Free unlimited access for 30 days limited time only.
Water or H2O H 2 O has the electron dot structure shown below. Step 4 Six valence electrons are used to attach the N atom to three I atoms. Thats one of Dr.
Get the detailed answer. So those two are a lone pair which is simply two electrons together.

Ncert Cbse Standard 12 Co Ordination Compounds Chapter 9 Inorganic Chemistry Chemistry Physics And Mathematics Ordination

25 8 2013 18 8 22 1 Ensenanza De Quimica Quimica Notas De Quimica

Lewis Dot Structures For Polyatomic Ions Clear Simple Practices Worksheets Chemistry Worksheets Worksheets