Brf3 Lewis Dot Structure
There are a total of 28 valence electrons for the BrF 3 Lewis structure. What is a Lewis structure or a dot structure.

Brf3 Lewis Structure Bromine Trifluoride In 2021 Lewis Chemical Formula Dots
The Lewis structure of BrF3 is started by writing Br in the center.

Brf3 lewis dot structure. The outermost valence electrons of the CH3I molecule must be understood while considering the Lewis structure of the molecule. BF3 has a total of 24 valence electrons which we have to set around the central atom. The electron dot structure of the CH3I molecule is also known as the CH3I Lewis structure.
As three electrons out of seven form a bond with the valence electrons in the Fluorine atom there are four nonbonding electrons on the central atom of BrF3. In the Lewis structure for BrF 3 there are a total of 28 valence electrons. Here in this post we described step.
Bromine and Fluorine both have 7 valence. BF3 Lewis Structure. For the BrF 3 Lewis structure calculate the total number of valence electrons for the BrF 3 molecule.
BrF3 Lewis Structure Molecular Geometry Hybridization and MO Diagram BrF3 known as Bromine Trifluoride is a fuming liquid consisting of inter-halogen combinations and bearing a pungent smell. Electron geometry and molecular geometry. BrF3 - Octahedral - Square pyramidal.
Drawing the Lewis Structure for BrF 3. The Br 3- Lewis structure has a total of 22 valence electrons. Bromine trifluoride - Wikipedia the free encyclopedia Bromine trifluoride is an interhalogen compound with the formula BrF 3.
Bromine is the least electronegative atom in the BrF 3 Lewis structure and therefore goes at the center of the. What is the formal charge of both oxygens. Electron geometry and molecular geometry.
Brf3 lewis dot structure Lewis structure for BrF3 - The QA wiki What is the Lewis structure of brF3. To know about BF3 Lewis structure we have to calculate the total number of valence electrons for the BF3 molecule. Bromine belongs to group 17 with 7 electron in the outermost shell.
In the BrO3- Lewis structure Bromine Br is the least electronegative and goes in the center of the dot structure. It determines the number of outermost valence electrons as well as the electrons engaged in the CH3I molecules bond formation. Each Bromine Br atom contributes 4 valence electrons and the negative sign - provides an additional valence electron for a total of 22.
Br is the central atom connected to each F atom by a single bond. For the BrF 3 Lewis structure youll need to put more than eight valence electrons on the Bromine atom. In the BrF 3 Lewis structure Bromine Br is the least electronegative atom and goes in the center of the Lewis structure.
Dots around the chemical symbol. Remember that Bromine Br can hold more than 8 valence electrons and have an expanded octet. A video explanation of how to draw the Lewis Dot Structure for Bromine Trifluoride along with information about the compound including Formal Charges Pola.
How many electrons does a single linebond represent in lewis dot structures. Be sure to put brackets and a - negative sign around the dot structure to show that Br 3- is an ion with negative one charge. Bromine trifluoride chemical formula is BrF3.
3 lone electron pairs will surround each F atom and 2. Going from left to right there are 6 electrons around O hence neutral 4 electrons around N hence positive and 7 electrons around the rightmost oxygens hence each is a formal anion. After determining how many valence electrons there are in BrF 3 place them around the central atom to complete the octets.
Both Br and F have seven valence electrons so the lewis structure will have a total of 28 electrons or 14 electron pairs. Three dashes are drawn from the Bromine atom with a Fluorine atom connected to each dash. Try structures similar to BF3 for more practiceON -O-_2 is the standard Lewis structure.
For the BrO3- Lewis structure calculate the total number of valence electrons for the BrO3-. Having a straw ie colorless to yellow appearance this chemical compound has several applications but also comes with a number of limitations and hazard issues. BrF3 has a Bent T shaped structure with Br as a central atom bonded with three F atoms three bond pairs and two lone pairs.
BrF3 does not follow the octet rule. Drawing BrF3 Lewis Structure is very easy to by using the following method. How many lone pairs are on the central atom of BrF3.
The Lewis structure of BrF3 will have three bonds between Br-F represented by lines and four nonbonding electrons represented as four dots on the Bromine atom. What is the formal charge of hydrogen. What is the formal charge of sulfur.
April 11th 2019 - This is the BrF3 Lewis structure We have a total of 28 valence electrons for the BrF3 Lewis structure Bromine is the least electronegative we ll put that at the center and then we ll put the Fluorines around the outside We ll put single bonds between the Fluorines and the Bromine That s six valence 2 4 6 valence electrons we ve used.

Vsepr Theory A Closer Look At Bromine Trifluoride Brf3 Henry Rzepa Vsepr Theory Theories Closer

Non Aqueous Solvents Nh3 Hf H2so4 N2o4 Pocl3 Socl2 Brf3 In 2021 Solvents Chemistry Agno

Hybridization Of Sf4 Sulfur Tetrafluoride In 2021 Molecules Chemical Formula Sulphur

Is C2h6 Polar Or Non Polar Ethane In 2021 Math Equations Chemical Formula Molecules

Is Nh2 Polar Or Non Polar Amide Ion In 2021 Nh 2 Molecules Electrons
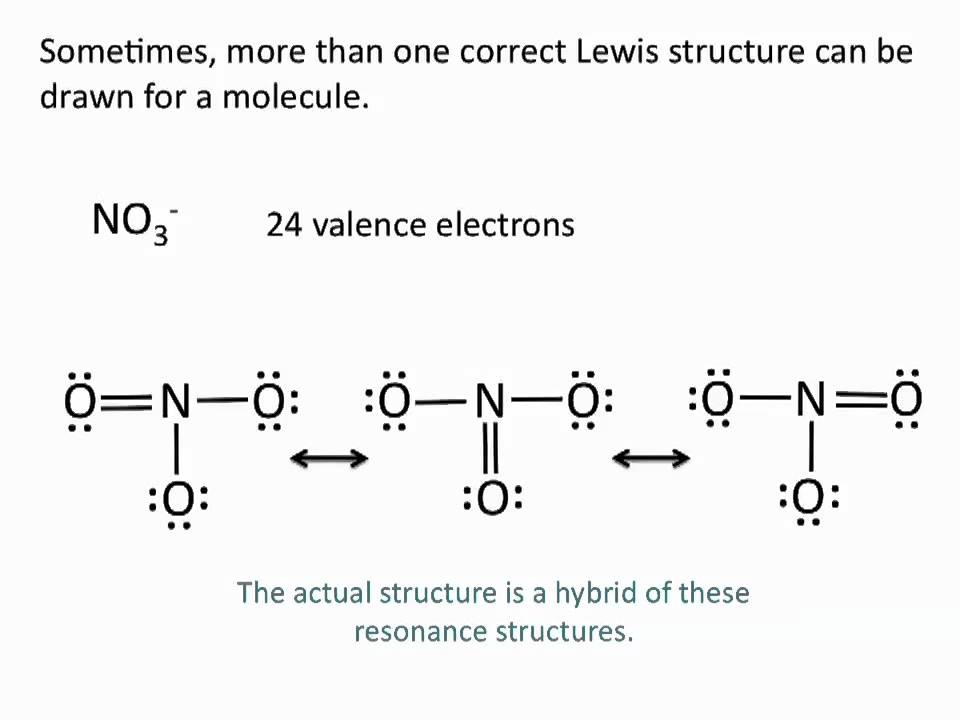
Drawing Lewis Structures Resonance Structures Chemistry Tutorial Youtube Chemistry Organic Chemistry Science Education

Hybridization Of Of2 Oxygen Difluoride In 2021 Molecules Oxygen Things To Come

Ccl4 Lewis Structure Carbon Tetachloride In 2021 Carbon Molecule Molecules Lewis

Ch3cl Lewis Structure Chloromethane In 2021 Lewis Molecules Methylation

Brf3 Lewis Structure Bromine Trifluoride In 2021 Lewis Chemical Formula Dots

Is Ch3oh Polar Or Nonpolar Methanol In 2021 Functional Group Molecules Chemical Formula

Is Hcn Polar Or Nonpolar Hydrogen Cyanide In 2021 Chemical Formula Molecules Polar

Ch4 Lewis Structure Methane In 2021 Lewis Methane Chemical Formula

Is Bf3 Polar Or Non Polar Boron Trifluoride In 2021 Boron Atom Molecules Chemical Formula

Ch4 Lewis Structure Methane In 2021 Lewis Methane Chemical Formula

Ccl4 Lewis Structure Carbon Tetachloride In 2021 Carbon Molecule Molecules Lewis


