Pf5 Lewis Acid Or Base
Why is PCl3 a Lewis base. Electron donating groups on an atom can increase the Lewis basicity of that atom while electron-withdrawing groups can increase the Lewis.
The phosphorus in PCl5 readily accepts electrons from other molecules.
Pf5 lewis acid or base. A Lewis acid is a species that can accept an electron pair whereas a Lewis base has an electron pair available for donation to a Lewis acid. It explains how to predict the products of a lewis acid-base. Ammonia acts as the Lewis base.
The answer to the first question is yes of course you can draw Lewis dot structures for both compounds recall P can have an expanded octet. Toxic and corrosive fumes are generated when this material is. The bisulfite ion is amphiprotic and can act as an electron donor or acceptor.
The answer to the second question is again yes they can both behave as Lewis acids again because of expanded octets and. CeSbF5 AsF5 PF5 One simple argument could be the size of the ceSb-center. It was introduced in the first half of the 20th century by Gilbert Newton Lewis.
Include geometry andformal charges in the structures. Why Cu2 is a Lewis acid. In a complex ion we have a central atom often consisting of a transition metal cation which acts as a Lewis acid and several neutral molecules or ions surrounding them called ligands that act as Lewis bases.
This product is also referred to as a Lewis adduct. Lewis acid is a compound or an ion which receives a pair of electron. By this definition every cation is an acid in addition to chemical compounds such as BF 3 and SiO 2.
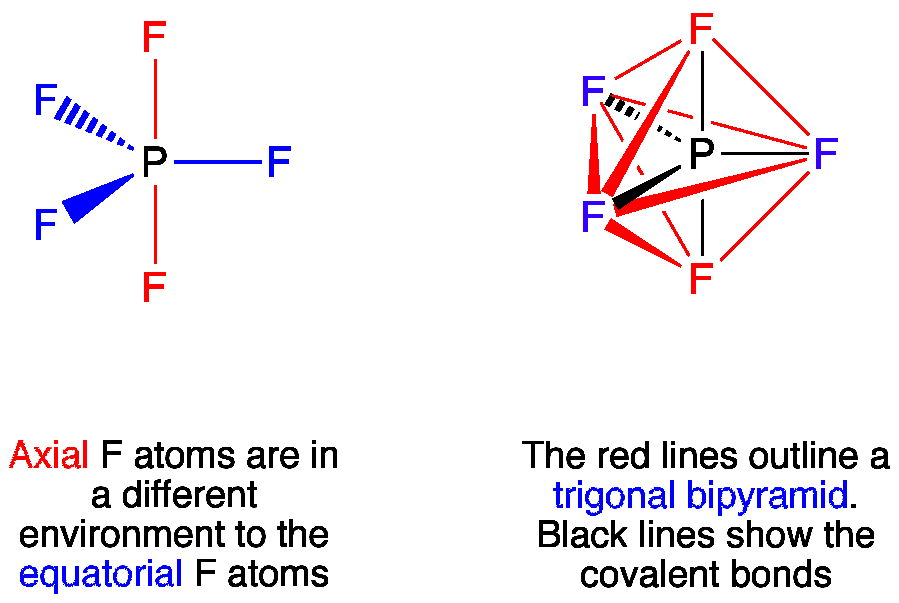
Lewis base is a compound or an ion which gives a pair of electron. It has 10 electrons five valence electrons fromphosphorus and five from five fluorine. B CH3 3 B OH 3.
It is extremely irritating to skin eyes and mucus membranes. I couldnt find a more precise explanation or rationalisation. Phosphorus pentafluoride is a colorless poisonous nonflammable compressed gas with a pungent odor.
Because it is bigger than for example a ceP-center it can accept more ligands. Because HF is a weak acid fluoride salts behave as bases in aqueous solution. Lewis acids A Lewis acid is an electron pair electrophilic that can attach electron pairs.
Therefore a Lewis base can donate a pair of electrons to a Lewis acid to form a product containing a coordinate covalent bond. Phosphorus trichloride has a lone pair and therefore can act as a Lewis base. An illustration detailing the reaction between a Lewis acid and base leading to.
Lewis acids and bases are described by the Lewis theory of acid-base reactions as electron-pair acceptors and electron pair donors respectively. Which compound will be a stronger Lewis acid PF5 or PCl5Explain briefly. My knowledge of the MO diagram of hypercoordinative compounds is really basic so.
This organic chemistry video tutorial provides a basic introduction into lewis acids and bases. This type of bond is usually represented by an arrow. Therefore it is considered as a Lewis acid.
The strength of the interaction between a Lewis acid and a Lewis base is controlled by at least two factors electronic and steric. Is PF5 an exception to the Lewis octet rule. CH4 acid or base.
Lewis acids which are electron pair acceptors and Lewis bases which are electron pair donors react to form adducts in which a coordinate covalent bond is formed. It explains how to predict the. I believe that thats correct where Arrhenius and Bronsted is a reaction with water but I believe that lewis acids and bases dont necessarily have to be in water.
Complex ions are examples of Lewis acid-base adducts. The best answer I can give is that there is enough space on ceSb in ceSbCl3 for an additional Lewis Base to interact with a nonbonding orbital on the ceSb atom. What is the adduct that isformed Include a picture if you can.
Among the Lewis acids Compounds with incomplete or unstable electron octet as. The Lewis definitions are an acid is a substance which can accept an electron pair from a base. Are you asking if you can draw Lewis dot structures or are you asking if these compounds can act as Lewis acids or bases.
For example with the Lewis acids BBr3 it forms a 11 adduct Br3BPCl3. Lewis acids and bases. The Lewis acid - base concept is a definition of acid and base which is independent of protons.
For example PF5 F- Pf6 is a lewis acid-base reaction. It is very toxic by inhalation and can cause pulmonary edema. Cu2 aq reacts with ammonia to form the complex ion CuNH342.
Generally the positive ions generally known as the cations and compounds which have incomplete d orbital non metalic oxide are lewis acid. A base is a substance which can donate an electron pair 24. While ceP can be hypervalent the LUMO in this case would be ceP-F sigmaast which is really high in energy meaning that it doesnt easily accept electron density from other species in solution.
How can these relative reactivities as a Lewis acids be rationalized. As a Lewis base F accepts a proton from water which is transformed into a hydroxide ion. Draw molecular geometries for the reactants and the product ofthe reaction of PF5 with the Lewis base NH3.
Solved The Uorides Bf3 Aif3 Sif4 And Pf5 Are Lewis Acids The All Form Very Stable Uoroanions When Treated With Lithium Uoride In Contrast The Course Hero

Incorrect Order Of Lewis Acid Character
Welcome To Chem Zipper Com Lewis Acid Base Concept

Molecular Geometry Of Pf5 Phosphorus Pentafluoride Youtube
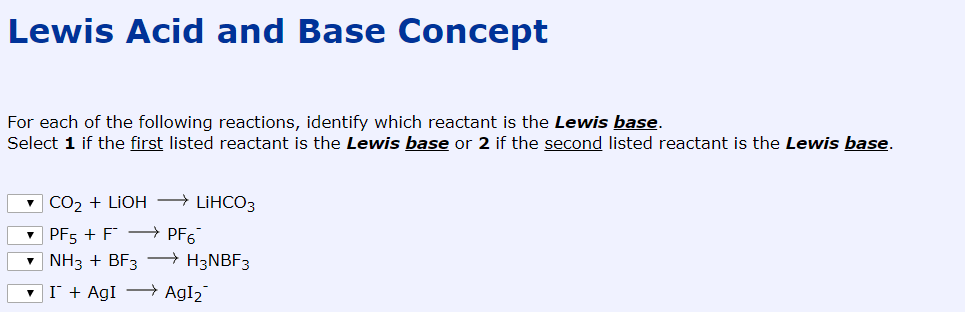
Lewis Acid And Base Concept For Each Of The Following Chegg Com
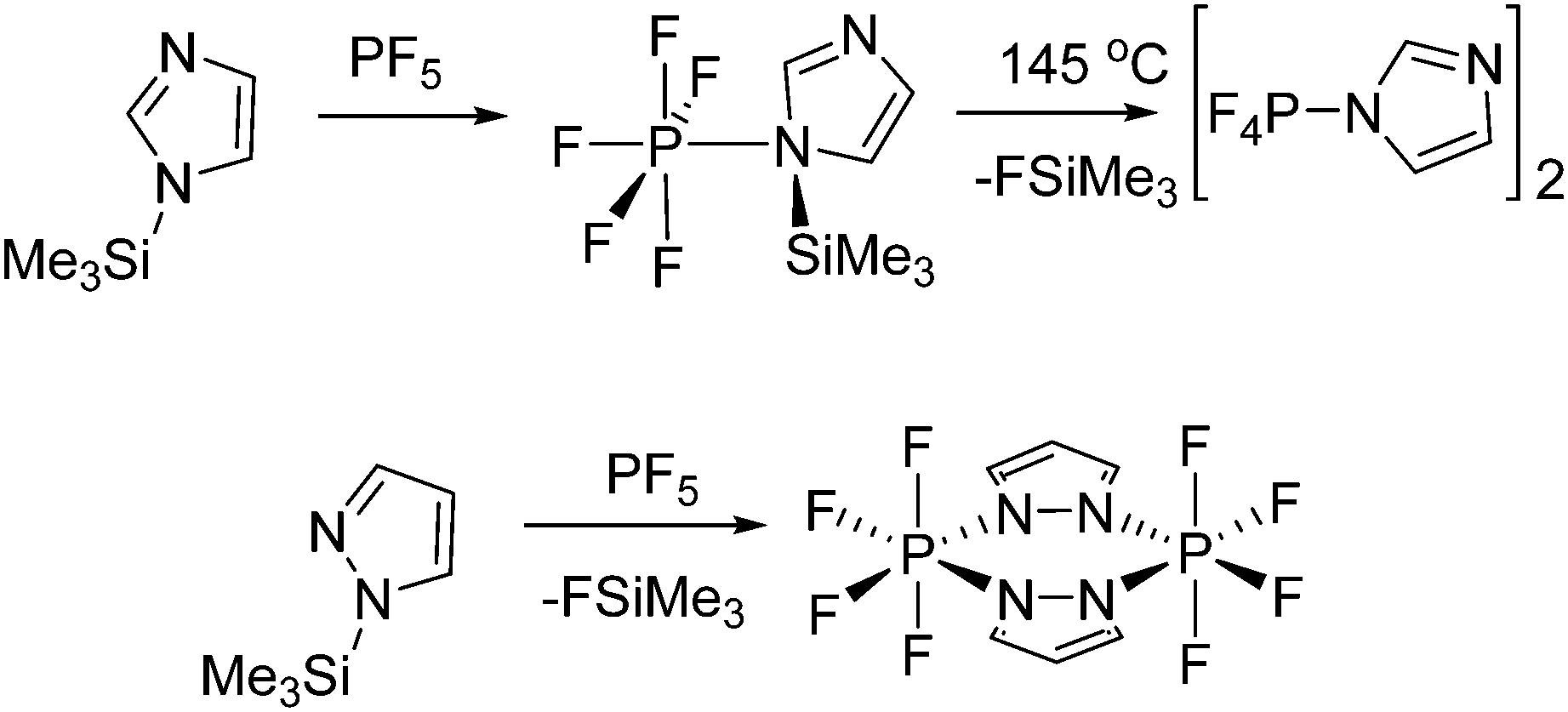
Phosphorus Lewis Acids Emerging Reactivity And Applications In Catalysis Chemical Society Reviews Rsc Publishing Doi 10 1039 C5cs00516g
Http Www Carbene De Wp Content Cm 1111 Tutorial 6 With Answers Pdf
Phosphorus Pentafluoride Wikiwand

Lewis Acid Catalyzed Synthesis Of Cyanidophosphates Blasing 2016 Chemistry A European Journal Wiley Online Library

Pf5 Lewis Structure Phosphorus Pentafluoride Youtube
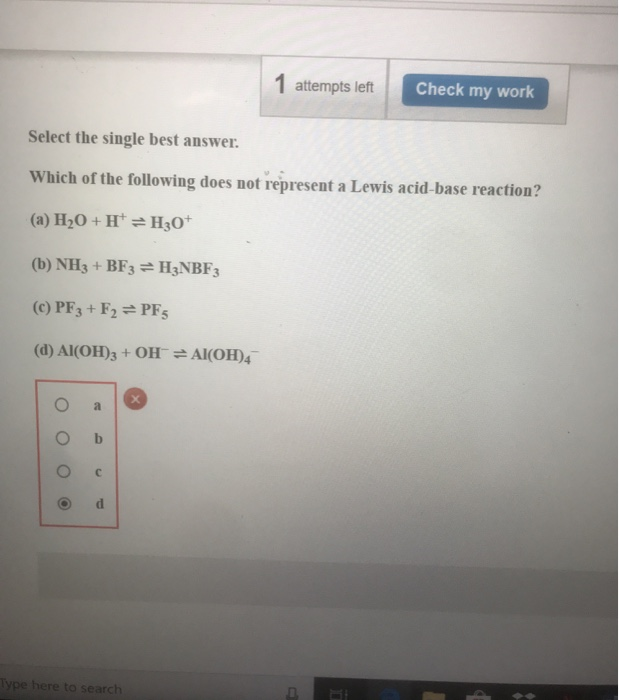
1 Attempts Left Check My Work Select The Single Best Chegg Com

Pf5 Lewis Structure And Molecular Geometry Made Easy Youtube
Https Www Neetprep Com Subject U3viamvjddo1na Topic Vg9wawm6njq4 Doubt Rg91ynq6mzk4njy
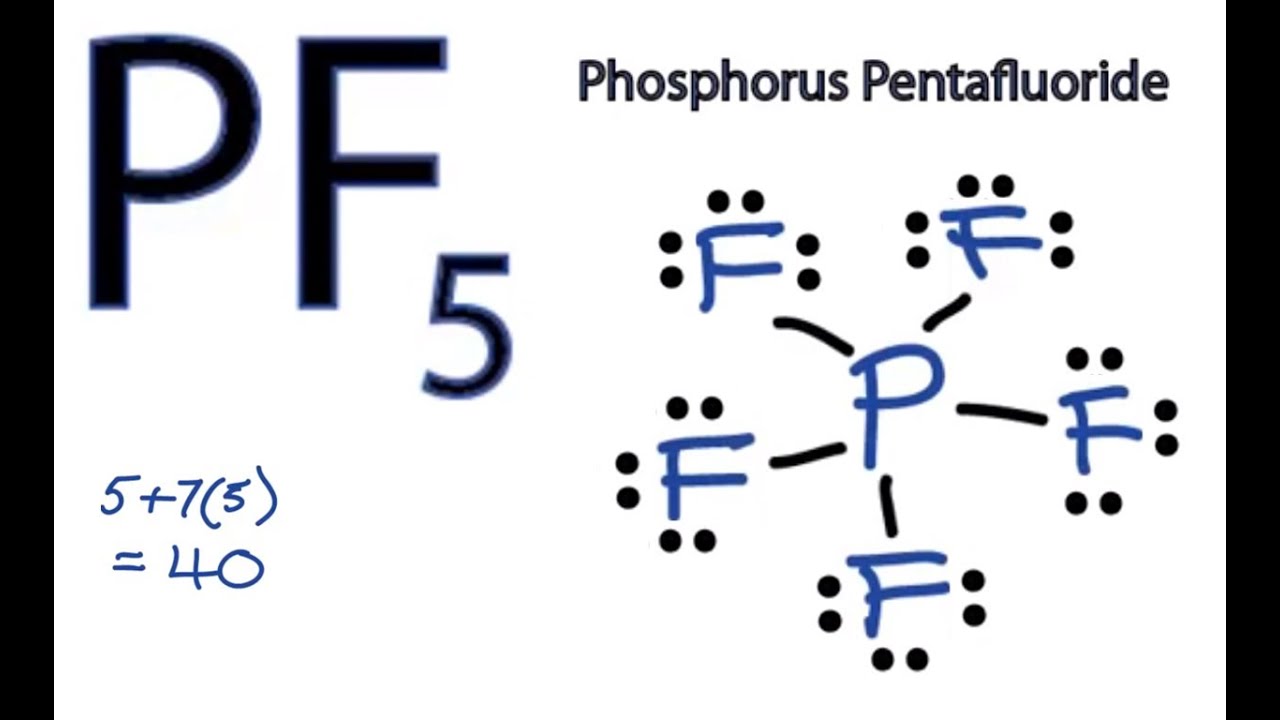
Pf5 Lewis Structure How To Draw The Lewis Structure For Pf5 Phosphorus Pentafluoride Youtube

Pf5 Lewis Structure How To Draw The Lewis Structure For Pf5 Phosphorus Pentafluoride Youtube

Vsepr Pf5 Phosphorus Pentafluoride

1 8 Chemistry 2810 Answers To Assignment 3 Topic Lewis


