Consider The Lewis Structure For Pcl3 Pcl3
Four outer atoms and zero lone pairs would be. Here Phosphorous 5 valence electrons Chlorine 7 valence electrons 3 Cl 73 21 So total valence electrons 26.
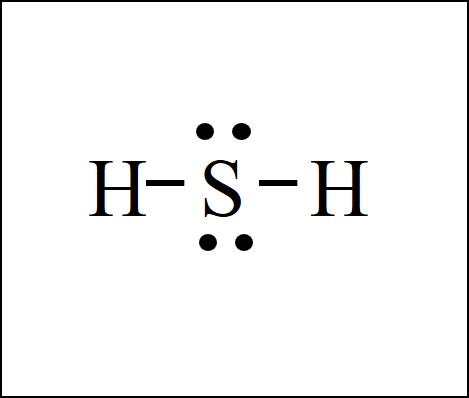
Lewis Dot Structure Easy Hard Science
PCl3 Lewis StructureLewis Structure of PCl3 Phosphorus TrichlorideDraw Lewis Structure for PCl3PClLewisStructureThis video will help you to draw the L.

Consider the lewis structure for pcl3 pcl3. Second place the valence electron on the iodine and hydrogen atoms. Central phosphorus atom obeysa. A step-by-step explanation of how to draw the PCl3 Lewis Dot Structure Phosphorus TrichlorideFor the PCl3 structure use the periodic table to find the tot.
For PCl 3 Total pairs of electrons are twenty 402 in their valence shells. Also there is a lone pair on phosphorus atom. SBr2 Lewis structure diagram in this phase.
Phosphorus trichloride has a lone pair and therefore can act as a Lewis base. What are the appropriate bond angles. Why is PCl3 a Lewis base.
Draw the Lewis structure for PCl3 phosphorus trichloride the chemical used commercially to prepare insecticides and flame retardants. 5 rows Phosphorus trichloride is made up of one Phosphorus atom and three Chlorine atoms having a chemical. Consider the Lewis structure for PCl3 what are the approximate bond angles.
There are requirements to be the center atom. When you draw a lewis structure selecting the center atom is very important because structure of the molecule depend on the center atom selection. Formula Lewis Structure Electronic Geometry Molecular Geometry Bond Angles Hybridization of Central Atom Bond Polarity Molecular Polarity yes or no CH HO NH4 Page 24 of 29 Explain the structure and geometry of SnCl2 draw a lewis structure.
PCl 3 lewis structure. Consider the lewis structure for pcl3. Now lets move on to the lewis structure of PCl3.
Number of single bonds number of double bonds. There is a lone pair on center phosphorus atom and each chlorine atom also has three lone pairs. Explain the structure and geometry of SnCl2 draw a lewis structure.
Why is the ability to draw a Lewis structure considered a core chemistry skill. Phosphorus trichloride has a lone pair and therefore can act as a Lewis base. Also there are no charges on atoms in PCl 3 lewis structure.
To draw the lewis structure first of all we need to sum up the valence electrons of all the atoms. First the valence electrons are placed around the carbon atom. In this post we discussed the method to construct the CH3I Lewis structure.
Now we need to consider a central atom. What is the molecular geometry of a molecule with 4 outer atoms and 1 lone pair on the central atom. We review their content and use your feedback to keep the quality high.
In this tutorial we will learn how to draw the lewis structure of PCl 3 with all theories. Selection of center atom of PCl 3. In this lewis structure of PCl 3 center phosphorus atom has made three single bonds with three chlorine atoms.
For example with the Lewis acids BBr3 it forms a 11 adduct Br3BPCl3. Incomplete octet or c. The electronegativity value in periodic groups grows from left to right in the periodic table and drops from top to bottom.
How_to_draw_a_lewis_dot_structure_for_pcl3 24 How To Draw A Lewis Dot Structure For Pcl3 DOC How To Draw A Lewis Dot Structure For Pcl3 Inorganic Chemistry-Gary Wulfsberg 2000-03-16 Both elementary inorganic reaction chemistry and more advanced inorganic theories are presented in this one textbook while showing the relationships between the two. The lewis structure of PCl3 can be explained as follows. In the Lewis structure of CH3I the formal charge on the terminal hydrogen atom is zero.
Draw a Lewis structure for PCl5 and answer the following questions based on your drawing For the Central phosphorus atomnumber of lone pairs. Experts are tested by Chegg as specialists in their subject area. The first step is to put six valence electrons around the sulfur atom as given in the figure.
Draw the Lewis structure for hydrogen the simplest molecule and explain the drawing. Who are the experts. For the PCl5 Lewis structure we first count the valence electrons for the PCl5 molecule using the periodic table.
Estimate the Cl-Sn-Cl bond angles. For example with the Lewis acidsBBr3 it forms a 11 adduct Br3BPCl3.

Pcl3 Lewis Structure Hybridization Molecular Geometry And Mo Diagram Techiescientist

Pcl3 Lewis Structure Lewis Structure Of Pcl3 Phosphorus Trichloride Draw Lewis Structure For Pcl3 Youtube

Cf2cl2 Lewis Structure How To Draw The Dot Structure For Cf2cl2 Dichl Drawings Dots Lewis

Pcl3 Lewis Structure Phosphorus Trichloride In 2021 Math Equations Lewis Chemical Formula
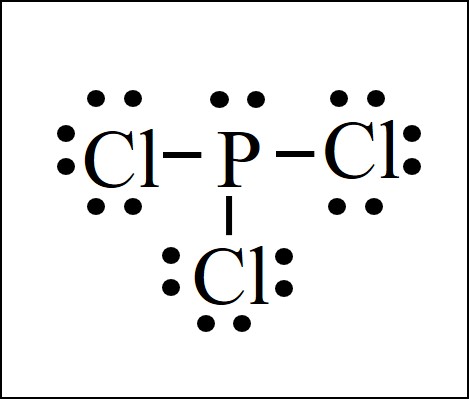
Lewis Dot Structure Easy Hard Science

Pcl3 Lewis Structure Phosphorus Trichloride In 2021 Math Equations Lewis Chemical Formula

How To Draw The Lewis Structure For Pcl4 Youtube

Is No3 Polar Or Nonpolar Nitrate In 2021 How To Find Out Molecules Polar

Is Nh2 Polar Or Non Polar Amide Ion In 2021 Nh 2 Molecules Electrons
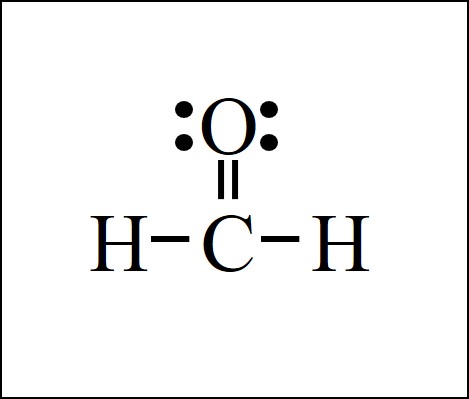
Lewis Dot Structure Easy Hard Science

Hybridization Of Of2 Oxygen Difluoride In 2021 Molecules Oxygen Things To Come

Becl2 Lewis Structure Beryllium Chloride In 2021 Math Equations Lewis Molecules

Lewis Structures For Covalent Molecules Step By Step Youtube
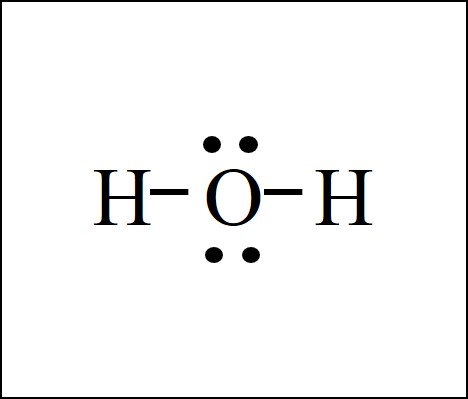
Lewis Dot Structure Easy Hard Science

Nacl Polar Or Nonpolar Sodium Chloride In 2021 Math Equations Molecules Sodium

Pcl3 Lewis Structure Hybridization Molecular Geometry And Mo Diagram Techiescientist

Nacl Polar Or Nonpolar Sodium Chloride In 2021 Math Equations Molecules Sodium

Ch2cl2 Lewis Structure Dichloromethane In 2021 Molecules Math Equations Hydrogen Atom

Http Www Youtube Com Watch V Qojoaosk5ui Lewis Chemistry Math Equations